Last Update 06 Jun 26
Fair value Decreased 21%IMDX: FDA Review Of Transplant Rejection Test Will Drive Bullish Repricing
Analysts have adjusted their price target for Insight Molecular Diagnostics from $10.50 to about $8.33, reflecting updated assumptions around revenue growth, profit margins, and future P/E expectations.
What's in the News
- Insight Molecular Diagnostics submitted its GraftAssureDx donor derived cell free DNA (dd cfDNA) assay kit to the FDA for regulatory review, following 12 years of clinical development and three years of focused product work. Source: company shareholder letter.
- The company reported that it is now engaged with 37 transplant centers in the U.S. and 11 internationally through its research use only assay and registry study, with 28 of the U.S. centers expressing interest in joining its registry trial. Source: company shareholder letter.
- Management indicated that the 37 U.S. transplant centers engaged with the company represent more than 25% of all transplants performed annually in the country and referenced a roughly US$2b addressable market for regulated transplant rejection testing. Source: company shareholder letter.
- Insight Molecular Diagnostics highlighted a new peer reviewed study in Transplant International that evaluated its GraftAssure technology in 249 kidney transplant patients across two independent cohorts, comparing dd cfDNA levels with histopathology based rejection indices. Source: Transplant International via company summary.
- The company reported that its GraftAssure Combination Model score outperformed fractional and absolute dd cfDNA measures alone in correlation analyses with several transplant health indices, and cited congress data indicating a positive predictive value above 80% at 25% prevalence using this score. Source: Transplant International study and World Transplant Congress 2025 data via company summary.
Valuation Changes
- Fair Value was reduced from $10.50 to about $8.33 per share, indicating a lower assessed valuation level for Insight Molecular Diagnostics.
- The Discount Rate was adjusted slightly from 7.13% to 7.11%, reflecting only a minor change in the required rate of return used in the model.
- Revenue Growth was revised from about 135.61% to about 123.87%, pointing to more conservative expectations for future sales expansion.
- The Profit Margin was nudged up from about 18.66% to about 19.04%, indicating a modestly higher assumed profitability on future revenues.
- The Future P/E was lowered from about 105.85x to about 95.94x, suggesting a reduced multiple applied to projected earnings in the updated valuation.
Key Takeaways
- Expanding clinical adoption and demographic trends are set to boost recurring revenue and gross margins through increased testing volume and broader market penetration.
- Strategic focus on high-margin digital PCR kits and new partnerships diversifies income sources, enhances profitability, and lessens dependence on external financing.
- Heavy reliance on a single customer, ongoing cash burn, and uncertain product adoption create substantial risks for revenue stability, profitability, and long-term market competitiveness.
Catalysts
About OncoCyte- Operates as a precision diagnostics company in the United States and internationally.
- Ongoing demographic shifts, including the global rise in organ transplants and aging populations, are expanding the total addressable market for advanced transplant rejection diagnostics, positioning OncoCyte to benefit from increased long-term testing volume and recurring revenue growth.
- Growing acceptance and adoption of precision medicine, along with recent claims expansions for high-frequency testing (e.g., 6 tests per patient in new screening protocols), could drive higher utilization rates of OncoCyte’s proprietary assays, leading to sustainable increases in revenue and gross margin as market penetration grows.
- Regulatory momentum and breakthrough device designation for companion drugs in antibody-mediated rejection (ABMR) is accelerating the clinical need for advanced monitoring tools like OncoCyte’s donor-derived cell-free DNA test, likely to improve payer coverage and support higher earning potential upon FDA clearance.
- The company's strategic shift toward high-margin, kitted molecular test products—supported by streamlined digital PCR workflows that are more cost-effective than legacy NGS methods—should improve net margins over time as the product mix evolves and direct-to-center adoption increases.
- Active expansion of partnerships and potential licensing agreements, evidenced by rising interest from large pharma and transplant centers in both transplant and oncology, provide new non-dilutive revenue channels that can underpin future earnings growth and reduce reliance on capital markets.
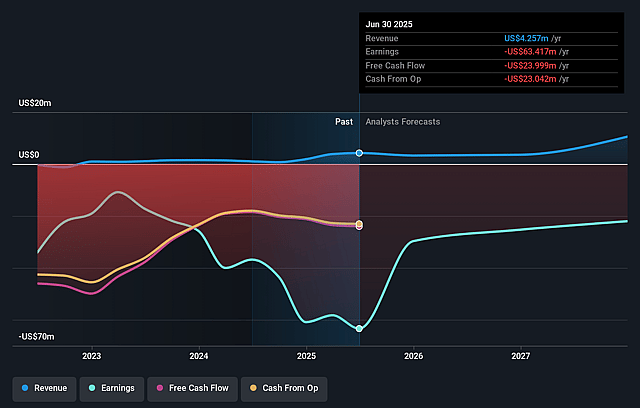
OncoCyte Future Earnings and Revenue Growth
Assumptions
How have these above catalysts been quantified?
- Analysts are assuming Insight Molecular Diagnostics's revenue will grow by 123.9% annually over the next 3 years.
- Analysts are not forecasting that Insight Molecular Diagnostics will become profitable in next 3 years. To represent the Analyst Price Target as a Future PE Valuation we will estimate Insight Molecular Diagnostics's profit margin will increase from -2454.6% to the average US Biotechs industry of 19.0% in 3 years.
- If Insight Molecular Diagnostics's profit margin were to converge on the industry average, you could expect earnings to reach $4.2 million (and earnings per share of $0.11) by about June 2029, up from -$47.8 million today.
- In order for the above numbers to justify the price target of the analysts, the company would need to trade at a PE ratio of 97.3x on those 2029 earnings, up from -3.5x today. This future PE is greater than the current PE for the US Biotechs industry at 16.6x.
- Analysts expect the number of shares outstanding to grow by 7.0% per year for the next 3 years.
- To value all of this in today's terms, we will use a discount rate of 7.11%, as per the Simply Wall St company report.
Risks
What could happen that would invalidate this narrative?- The company’s substantial dependency on a single large pharma customer for the majority of Q1 revenue highlights significant customer concentration risk and unpredictable revenue streams, which may disrupt stable long-term earnings.
- Persistent negative cash flow and a quarterly cash burn rate of ~$6 million, coupled with a lack of near-term commercial revenue, indicate ongoing reliance on external financing or capital markets, increasing dilution risk and pressuring future net margins and shareholder value.
- The clinical and commercial success of core products is highly contingent on achieving FDA approval and rapid physician adoption; delays, resistance to changing physician behavior, or slower switch-over by transplant centers could materially impact projected revenue growth and prolong path to profitability.
- The company is highly focused on a limited set of diagnostic products and is in an early commercialization stage; any underperformance or failure to gain reimbursement (e.g., delays with MolDX for DetermaIO) could materially constrain earnings and hamper sustainable revenue generation.
- Technological innovation in the molecular diagnostics sector is rapid and competitive; larger, better-financed competitors or next-generation platforms (NGS, multi-omics) may outpace OncoCyte’s offerings, eroding potential market share and putting long-term revenue and gross margin expansion at risk.
Valuation
How have all the factors above been brought together to estimate a fair value?
- The analysts have a consensus price target of $8.33 for Insight Molecular Diagnostics based on their expectations of its future earnings growth, profit margins and other risk factors.
- However, there is a degree of disagreement amongst analysts, with the most bullish reporting a price target of $12.0, and the most bearish reporting a price target of just $4.0.
- In order for you to agree with the analysts, you'd need to believe that by 2029, revenues will be $21.9 million, earnings will come to $4.2 million, and it would be trading on a PE ratio of 97.3x, assuming you use a discount rate of 7.1%.
- Given the current share price of $5.24, the analyst price target of $8.33 is 37.1% higher.
- We always encourage you to reach your own conclusions though. So sense check these analyst numbers against your own assumptions and expectations based on your understanding of the business and what you believe is probable.
Have other thoughts on Insight Molecular Diagnostics?
Create your own narrative on this stock, and estimate its Fair Value using our Valuator tool.
Create NarrativeHow well do narratives help inform your perspective?
Disclaimer
AnalystConsensusTarget is a tool utilizing a Large Language Model (LLM) that ingests data on consensus price targets, forecasted revenue and earnings figures, as well as the transcripts of earnings calls to produce qualitative analysis. The narratives produced by AnalystConsensusTarget are general in nature and are based solely on analyst data and publicly-available material published by the respective companies. These scenarios are not indicative of the company's future performance and are exploratory in nature. Simply Wall St has no position in the company(s) mentioned. Simply Wall St may provide the securities issuer or related entities with website advertising services for a fee, on an arm's length basis. These relationships have no impact on the way we conduct our business, the content we host, or how our content is served to users. The price targets and estimates used are consensus data, and do not constitute a recommendation to buy or sell any stock, and they do not take account of your objectives, or your financial situation. Note that AnalystConsensusTarget's analysis may not factor in the latest price-sensitive company announcements or qualitative material.

